Effects of COVID-19 on Syringe Service Programs
People who use drugs (PWUD) are among the most vulnerable populations for COVID-19.
They have increased prevalence of pre-existing co-morbidities that may increase their risk of a negative outcome following COVID-19 exposure, for example respiratory and cardiovascular diseases due to smoking cigarettes, marijuana, or other substances. Syringe service programs (SSPs) not only provide sterile injection equipment to PWUD, but also other health services such as HIV and hepatitis C (HCV) testing, referrals to substance use treatment, and overdose education and naloxone distribution. SSPs often serve as a primary point of healthcare access for PWUD. Recent publications by former CHERISH Pilot Grant Recipient Tyler Bartholomew and by Sara Glick from the University of Washington Department of Medicine describe the effect of COVID-19 on services provided by SSPs in the United States.

Tyler Bartholomew, PhD
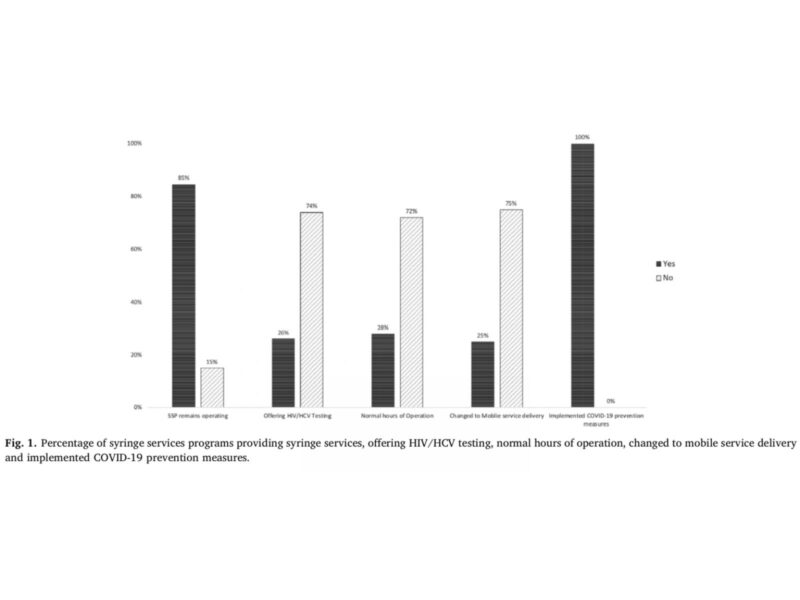
Bartholomew and colleagues conducted a national telephone survey in March of 2020 with 65 SSPs from 33 states to collect information. In this survey, SSP staff were asked to describe operational changes, changes to HIV and HCV testing services, and precautions to protect staff. Ten of the surveyed SSPs (15.4%) located across 9 states discontinued all SSP operations, sixteen (24.6%) switched to mobile delivery exclusively, and only seventeen (26.1%) continued to provide HIV and HCV testing.
These findings were consistent with those reported from a mixed methods study conducted by Sara Glick and colleagues, including CDUHR Investigator Don Des Jarlais from New York University College of Global Public Health. They found in a short survey administered by the North American Syringe Exchange Network (NASEN) conducted in the first two weeks in April 2020 that 43% of the 173 SSPs responding decreased service provision due to COVID-19, including medication for opioid use disorder and HIV and HCV testing and treatment services. They also found that approximately one quarter of SSPs closed sites due to COVID-19 related concerns such as staff safety and staff shortage.
More than a quarter of the sites also surveyed by NASEN reported conducting COVID-19 testing onsite for PWUD. This highlights the opportunity for SSPs to become a source for COVID screening and surveillance of this vulnerable population. During qualitative interviews with SSP staff in five COVID-19 hotspots, Glick and colleagues found that SSPs were not included in emergency planning in many states, and were not considered essential businesses. Qualitative interviews also confirmed that HIV and HCV testing decreased or stopped, that syringe and naloxone distribution were prioritized, and that demand for services remained high.