
Announcing the 2025-2026 CHERISH Pilot Grant Recipients
The CHERISH Pilot Grant & Training Core is pleased to support five early-career researchers in their academic journey leading pilot investigations on healthcare utilization, health outcomes, and health-related behaviors related to the care of people who use substances. The funded projects respond to CHERISH’s overall mission and topics of special interest in the call for […]

Health Economic Outcomes of a Minimal Monitoring Approach to Providing HCV Therapy
In a new article in Hepatology Communications, lead author and CHERISH Population Data & Modeling Core Director Benjamin Linas and colleagues report new economic analyses from MINMON, an AIDS Clinical Trial Group (ACTG 5360) study that used a minimal monitoring approach to provide treatment for chronic hepatitis C virus (HCV). Findings suggest that the simplified […]

Simulation Consultation: San Francisco Department of Public Health
The Syndemics Lab, led by CHERISH Population Data & Modeling Core Director Benjamin Linas, uses simulation models to investigate population-level outcomes, costs, and cost-effectiveness of interventions and care delivery models to treat HIV, hepatitis C virus (HCV), and substance use disorder. In addition to their core research, the Boston-based team collaborates with real-world decision-makers to […]

Hepatitis C Virus Screening in Pregnant and Nonpregnant Women After Universal Screening Guidelines
Hepatitis C virus (HCV) testing guidelines were updated in 2020 to provide universal, one-time screening for adults ages 18 through 79 and during each pregnancy. This departure from their 2013 recommendation not only aims to improve early detection of HCV across age groups but also acknowledges pregnancy as a critical period for diagnosing and treating […]
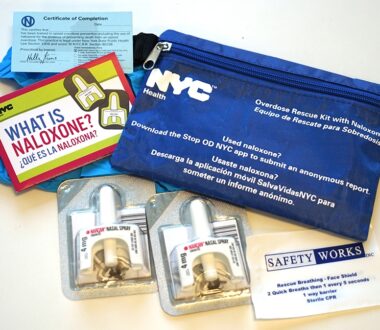
Research Highlights Prevention Efforts in Fentanyl Overdoses
The overwhelming majority of those in New York City who obtained a naloxone kit to counteract opioid overdose had a high need for the drug, according to a new study by Weill Cornell Medicine investigators and the New York City Department of Health and Mental Hygiene. The study, published Jan. 16 in the Journal of Urban Health, […]

Offering Buprenorphine in Homeless Shelters in Massachusetts Projects Life- and Cost-saving Results
Overdose is the leading cause of death among people with opioid use disorder (OUD) who are experiencing homelessness. Innovative care models that meet this population outside of office-based settings can improve access to buprenorphine, a medication for treating OUD, and avert fatal overdoses. In this study, Avik Chatterjee, lead author and addiction specialist at Boston […]

We’re barely using a key resource to help people with addiction
Rachel was making her 25th visit to the emergency department. The wound on her leg from injecting drugs had spread to her entire calf and required a lengthy course of antibiotic treatment in the hospital. The few times she had been admitted to the hospital, she had left without finishing treatment because the pain and […]

Catching Up with Pilot Grant Recipients Sierra Castedo de Martell and Bulat Idrisov
In preparation for the 11th cycle of CHERISH pilot grants that is now open, former pilot grant recipients Sierra Castedo de Martell and Bulat Idrisov joined CHERISH Pilot Grant Director Brandon Aden, Senior Research Program Manager Jared Leff, and Communications Manager Bonnie Tse online to meet prospective applicants. Attendees raised questions about eligibility, the pilot […]

Cost-Effectiveness of Fibrosis Staging
More than 4 million people in the U.S. live with a hepatitis C virus (HCV) diagnosis. However, as of 2022, fewer than one-third of individuals infected with HCV have been cured due to barriers related to high direct-acting antiviral (DAA) pricing and the steps involved in determining someone’s degree of liver disease or fibrosis. In […]

A Cost-Effectiveness of Implementation Facilitation to Promote Emergency Department–Initiated Buprenorphine for Opioid Use Disorder
In the last eight years, emergency departments (EDs) have seen an influx of visits related to opioid use disorder (OUD). For the same reason, researchers have been citing EDs to be a critical setting and reachable moment for initiating OUD medications, the gold standard of care. Buprenorphine is advantageous as a low-barrier OUD treatment compared […]

Racial and Ethnic Disparities Along the Hepatitis C Care Cascade Among Priority Populations
Over the last two decades, injection opioid use has increased among reproductive-aged women, with resultant increases in mother-to-child hepatitis C virus (HCV) transmissions in the U.S. Direct-acting antiviral (DAA) treatment is highly effective at reducing and eliminating the risk of HCV transmissions. Yet, studies show lower odds of receiving DAA treatment for women compared to […]

Announcing the 10th Pilot Grant Cohort Advancing the Treatment of Substance Use Disorders, HCV, and HIV
CHERISH is excited to welcome a new pilot grant cohort for the 10th year. Spearheaded by CHERISH Pilot Grant Program Director Brandon Aden, the program is designed to help researchers gain familiarity with health economic evaluations, apply related methodologies to their pilot research, and build out the next stage of their careers. This year, CHERISH […]
Engage with CHERISH
Submit a Consultation Request or Contact Us to learn more about how CHERISH can support your research or policy goals.